Understanding when a generic or biosimilar opportunity truly opens is rarely straightforward. Regulatory exclusivity and patent protection follow different timelines, and confusing them can lead to costly strategic mistakes.
For companies planning generic (ANDA/Para IV) or biosimilar (aBLA) development, understanding how these timelines interact is critical. Misinterpreting them can delay development planning, affect filing strategies, and cause companies to miss valuable market entry opportunities.
This article breaks down the fundamentals by explaining:
- The difference between NCE/NBE regulatory exclusivity and patents
- How approval timelines translate into filing windows
- Why tracking these milestones helps companies identify Para IV and biosimilar opportunities earlier
Regulatory Exclusivity vs Patent Protection: Two Different Systems
One of the biggest misconceptions in pharmaceutical development is assuming that patent protection and regulatory exclusivity are the same thing. In reality, they operate under completely different frameworks.
Regulatory Exclusivity
Regulatory exclusivity is granted by the FDA at the time of drug approval. It prevents competing companies from submitting certain types of applications for a defined period.
Typical examples include:
- New Chemical Entity (NCE) Exclusivity: 5 years
- New Biological Entity (NBE) Exclusivity: 12 years
- Orphan Drug Exclusivity: 7 years
Exclusivity exists to reward innovation and give the original sponsor a period of market protection.
However, exclusivity does not depend on patents and runs independently of them.
Patent Protection
Patents are granted by the national or regional government patent offices, such as the Indian Patent Office (IPO), European Patent Office (EPO), or US Patent and Trademark Office (USPTO), not the FDA.
They protect intellectual property related to the drug and may include:
- The active molecule
- Formulations
- Manufacturing processes
- Methods of use
- Delivery mechanisms
Unlike regulatory exclusivity, a single product may have multiple patents with different expiry dates.
This is why the patent landscape for a drug often extends far beyond its initial approval.
Visualizing How Exclusivity and Patents Interact
Understanding the interaction between regulatory exclusivity and patent protection becomes easier when viewed as a timeline.
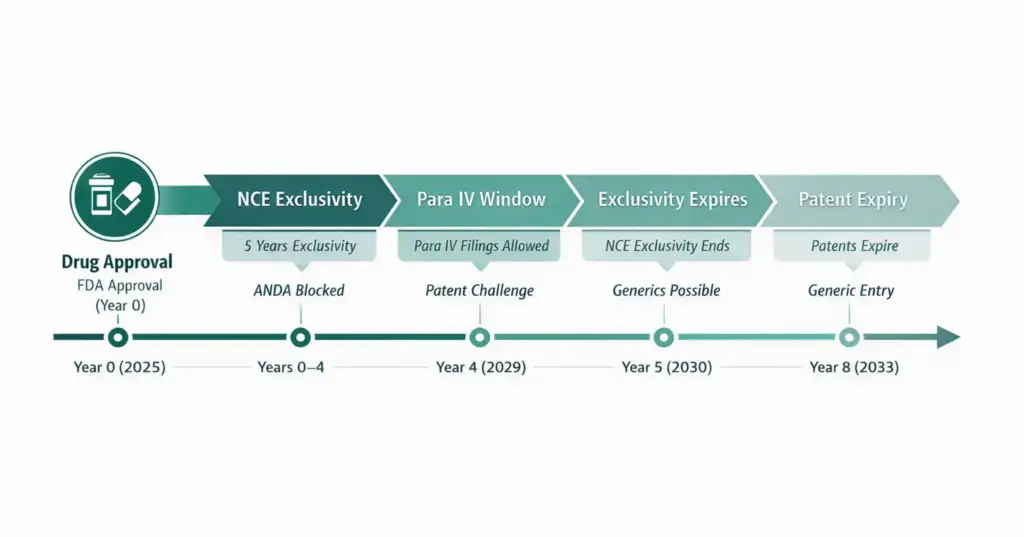
Figure 1: NCE timeline showing drug approval → exclusivity period → Para IV filing window → exclusivity expiry → patent expiry.
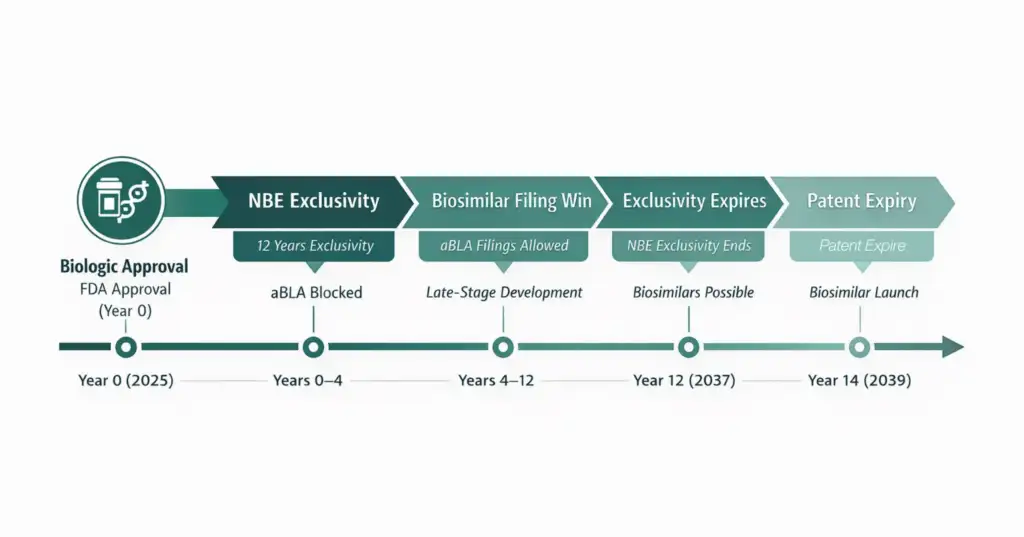
Figure 2: NBE timeline showing biologic approval → NBE exclusivity →biosimilar filing window → exclusivity expiry → patent protection period.
Even after regulatory exclusivity ends, patents may still delay market entry unless they expire or are successfully challenged.
How NCE and NBE Timelines May Differ
| Timeline Event | NCE Example | NBE Example |
| Drug receives FDA approval | 2025 | 2025 |
| Regulatory exclusivity active | 2025–2029 (5 years) | 2025–2037 (12 years) |
| First ANDA / Para IV or aBLA filing possible | 2029 (Year 4) | 2029 (Year 4) |
| Regulatory exclusivity expires | 2030 (Year 5) | 2037 (Year 12) |
| Key patent expiry | 2033 | 2039 |
Key insight:
Even after exclusivity ends, patent protection may still delay generic or biosimilar entry unless patents expire or are successfully challenged.
Where Patent Expiry Fits In
Patent timelines often overlap with regulatory exclusivity, but they do not necessarily align.
Three common scenarios occur:
1. Patent Expires After Exclusivity
If the primary patents extend beyond regulatory exclusivity, generic or biosimilar entry may still be delayed.
2. Patent Expires Before Exclusivity
Even if patents expire earlier, regulatory exclusivity may still prevent filing or approval.
3. Patent Challenges (Para IV)
A generic developer may challenge the innovator’s patents by filing an ANDA with a Paragraph IV (Para IV) certification, asserting that the listed patents are either invalid, unenforceable, or will not be infringed by the proposed generic product.
This is one of the primary pathways through which companies attempt to enter the market before patent expiry.
Once a Para IV filing is submitted, the innovator company may initiate patent litigation, which plays a key role in determining the timing of generic entry. If the innovator sues within 45 days of a Para IV filing, FDA approval of the generic is automatically stayed for up to 30 months, unless the case is resolved sooner.
Several outcomes are possible:
First-to-File Advantage
The first generic applicant to file a Para IV certification may be eligible for 180-day exclusivity, allowing it to be the sole generic competitor for a limited period after launch.
Successful Patent Challenge
If the court rules in favor of the generic company, it may receive approval to launch its product before the original patent expiry, creating an early market entry opportunity.
Settlement Between Parties
In many cases, innovator and generic companies reach a settlement agreement, where the generic is allowed to launch at a negotiated future date, often before full patent expiry but after a defined period.
Litigation Outcome Favors Innovator
If the innovator successfully defends its patents, generic entry may be delayed until patent expiry.
When Multiple Generic Players File ANDAs
In many cases, multiple generic companies monitor the same regulatory milestones and prepare to file their ANDA applications as soon as the filing window opens.
This often creates a highly competitive situation.
For example:
- First applicants may qualify for 180-day exclusivity.
- Multiple companies may submit Paragraph IV ANDA filings on the same day. In such cases, the 180‑day exclusivity is shared among the first filers.
- Patent litigation between innovators and generic applicants determines who ultimately launches first
As a result, companies closely track approval dates, exclusivity timelines, and patent landscapes to align development programs with potential filing opportunities.
Key FDA Guidance Points
- First Applicant Definition: Any ANDA filer who submits a complete Paragraph IV certification on the first day qualifies.
- Shared Exclusivity: When more than one filer qualifies, exclusivity is not divided sequentially — it runs concurrently for all first filers.
- Triggering Exclusivity: The exclusivity period begins when one of the first filers starts commercial marketing.
- Litigation Impact: Patent disputes or settlements still determine which companies can actually launch during the exclusivity window.
Why the 4-Year NCE Filing Window Is Closely Watched
For drugs granted NCE exclusivity, the four-year mark after approval represents a critical milestone for generic developers.
ANDA submissions referencing the innovator product are generally blocked for the first four years. After that, companies may submit Paragraph IV certifications challenging the listed patents.
This makes the Year 4 filing window one of the most closely monitored timelines in generic drug development.
Generic companies often begin development programs years in advance to ensure they are ready to file when this window opens.
Being among the earliest filers can provide a competitive advantage, particularly because the first successful Paragraph IV applicant may receive 180-day exclusivity.
Turning Regulatory Timelines Into Strategic Opportunity
Understanding how NCE/NBE exclusivity and patent protection interact allows companies to better plan development strategies and identify potential market opportunities.
Tracking these milestones helps organizations:
- anticipate generic and biosimilar development opportunities
- prepare for regulatory filing windows
- align development timelines with market entry strategies
- secure comparator drugs required for clinical and bioequivalence studies
Spring Bio Solution supports pharmaceutical and biotech companies in this process by helping development teams:
- track new drug approvals and exclusivity timelines
- monitor regulatory opportunity windows
- source comparator drugs for clinical programs through compliant global supply channels
This allows companies to align their development strategies with evolving regulatory timelines.
FAQs:
Can generics launch immediately after NCE exclusivity expires?
Not necessarily. While exclusivity may end, patents can still prevent market entry unless they expire or are successfully challenged.
How do biosimilar timelines differ from small-molecule generics?
Biologics receive 12 years of regulatory exclusivity, significantly longer than small-molecule drugs. Biosimilar developers typically plan development programs years in advance to align with these timelines.
Why do multiple generic companies file ANDAs at the same time?
Companies monitor the four-year NCE filing window closely, often preparing their applications years in advance to compete for first-to-file advantages.
Why is tracking new drug approvals important?
Tracking approvals helps companies identify:
– Potential generic opportunities
– Biosimilar development windows
– Upcoming regulatory filing timelines
Early visibility helps companies align development programs strategically.
What role does Spring Bio Solution play in this ecosystem?
Spring Bio Solution supports pharmaceutical and biotech companies by tracking drug approvals and exclusivity timelines, identifying development opportunities, and sourcing comparator drugs for clinical trials and bioequivalence studies—helping teams stay aligned with regulatory timelines.





